Medical Optical Fiber Technology by OFS
With more than 35 years of experience in designing and manufacturing fiber optic products and solutions for the medical industry, OFS today continues to innovate in this area, working closely with its customers to help bring life-saving, life-enhancing medical technology to market.
From the glass preform to the optical fiber and cable design to the finished probe assembled in a low-bioburden room, OFS’ engineering and manufacturing expertise help make vision and custom design a reality.
Dedicated to Medical Standards
OFS manufacturing facilities and quality management systems ensure optimal performance and peace of mind for our partners.
- ISO 13485 certified
- FDA Good Manufacturing Practices
- Low-bio-burden assembly rooms
- ISO10993 for biocompatibility and traceability
- Biocompatibility testing to support customers
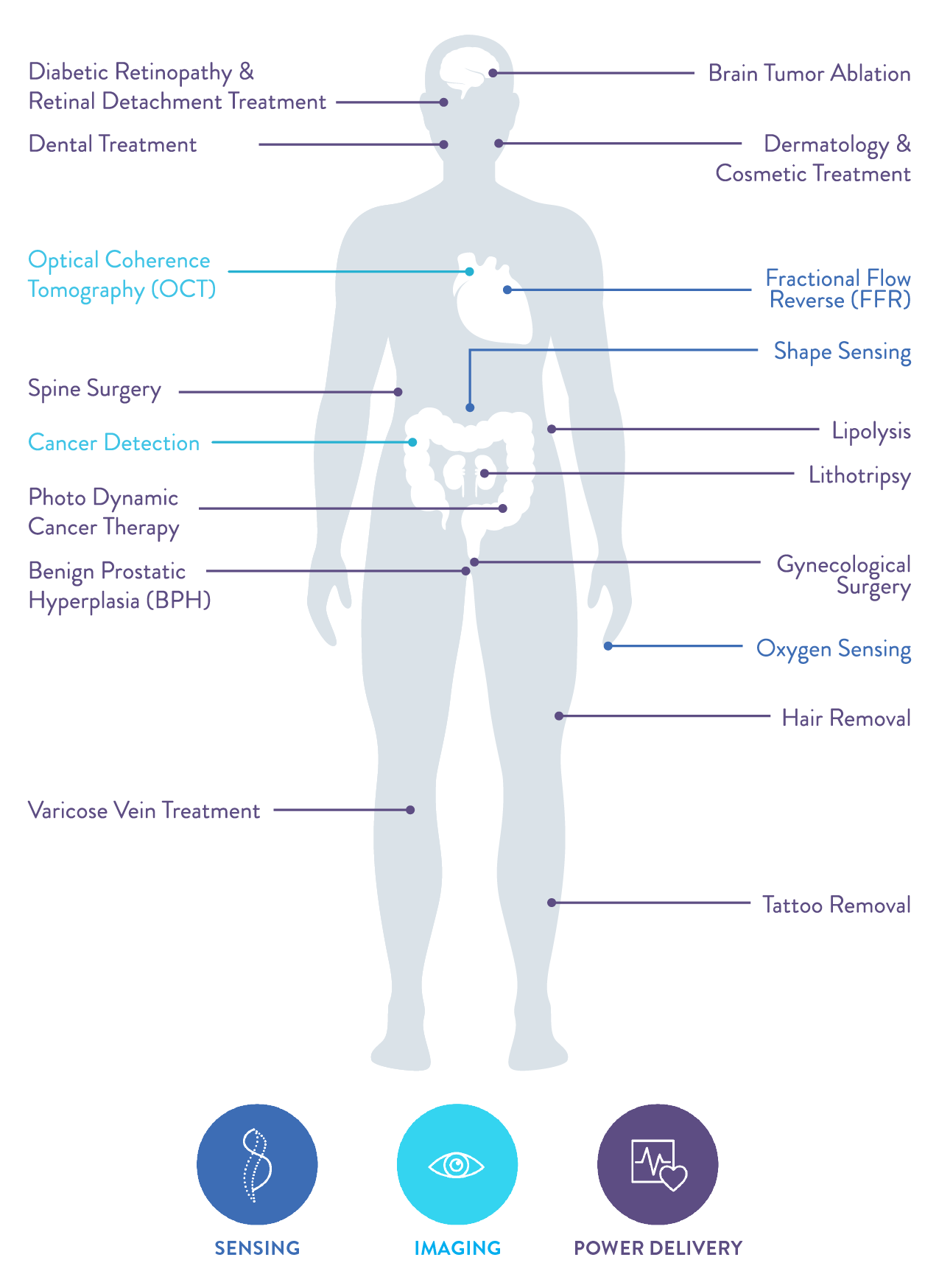
Medical Applications We Support